The issue
What is antibiotic resistance?
Antibiotic resistance—or antibioresistance—is defined as the ineffectiveness of antibiotic treatment against the targeted bacterial infection.
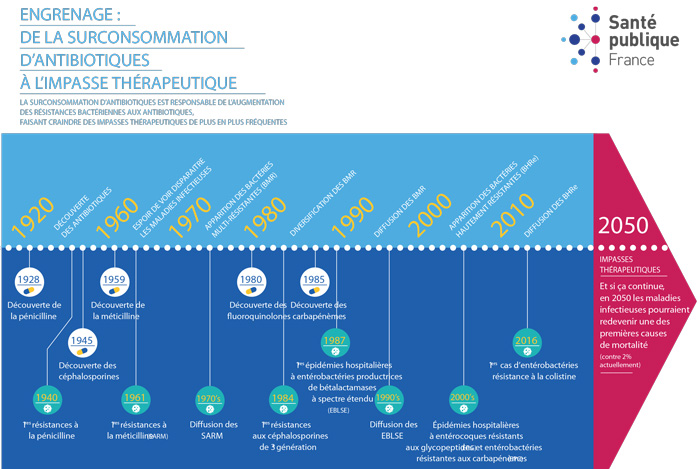
The repeated administration of antibiotics in humans or animals is responsible for the increase in bacterial resistance to antibiotics: the presence of antibiotics in the body promotes the selection of bacteria that are resistant, either naturally, through mutation, or through the exchange of genetic material (plasmids) with other bacteria. Absorbed antibiotics eliminate susceptible bacteria, leaving room for resistant bacteria. These can be transmitted and spread among individuals, particularly in healthcare settings. They make subsequent antibiotic treatments less effective for both the patient and the community.
Antibiotic resistance has gradually developed and now affects all pathogenic bacteria. It results from the repeated administration of antibiotics in humans or animals, which creates conditions—known as “selective pressure”—that promote the emergence and spread of antibiotic-resistant strains. Today, new antibiotics are rare, and it is sometimes difficult, if not impossible, to treat certain infections.
Antibiotic resistance results either from chromosomal mutations (changes in existing genes) or from the integration of small circular DNA molecules that are transmitted from bacterium to bacterium (plasmids). Chromosomal resistance affects only one antibiotic or one family of antibiotics at a time. Plasmid-mediated resistance is the most common (accounting for 80% of acquired resistance) and can affect multiple antibiotics, or even multiple families of antibiotics. This is referred to as multidrug resistance. The transfer of resistance mechanisms can occur from one strain to another or from one species to another. The accumulation of resistance mechanisms within a single bacterial strain can lead to treatment dead-ends. There are numerous resistance mechanisms: production of an enzyme that breaks down the antibiotic, impermeabilization of the bacterial cell wall, modification of the antibiotic’s target, and so on.
Key statistics on antibiotic resistance in human health

What causes antibiotic resistance?
Inappropriate use of antibiotics
Antibiotics are effective only against bacterial infections. They are still too often prescribed for viral infections such as the flu. The repeated administration of antibiotics in humans or animals is responsible for the rise in bacterial resistance to antibiotics by creating what is known as “selection pressure”: the level of antibiotic in the body promotes mutations and plasmid exchanges responsible for the acquisition of antibiotic resistance. This phenomenon tends to eliminate susceptible bacteria, leaving room for resistant bacteria. The more antibiotics are taken, the greater the risk of resistant bacteria emerging. These resistant bacteria make subsequent antibiotic treatments less effective for both the patient and the community.
How can we combat antibiotic resistance?
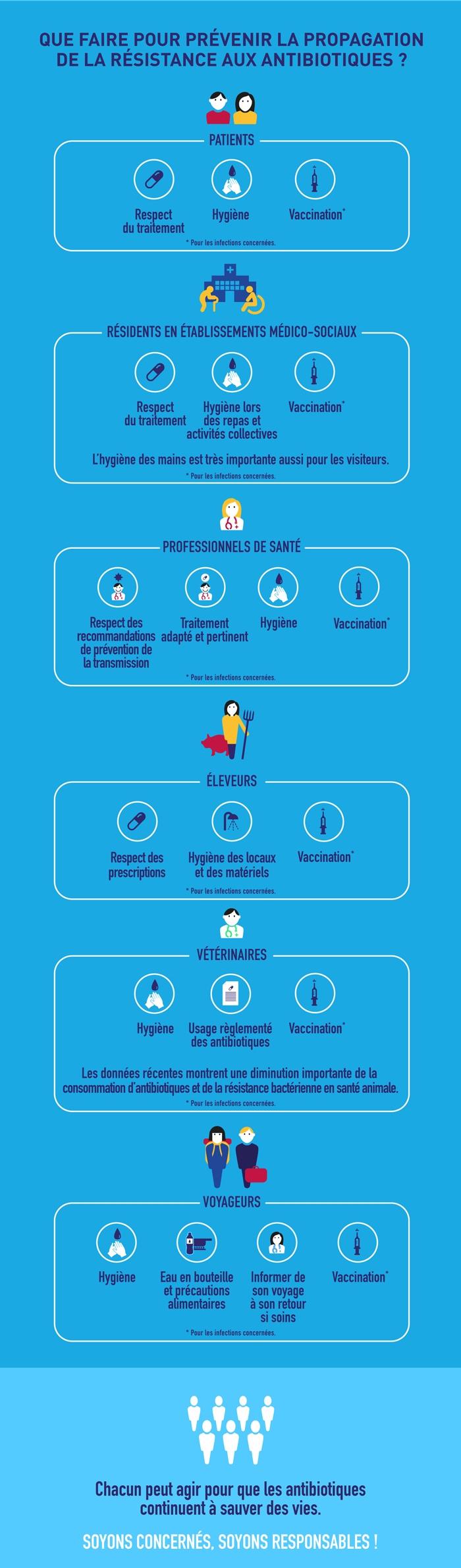
Controlling antibiotic resistance requires: better use of antibiotics to reduce selection pressure and the adoption of infection control measures to limit the spread of bacterial infections.
Better use
To ensure antibiotics remain effective—that is, to limit the emergence of resistant bacteria—certain principles of use must be followed: the right drug, at the right dose, and for the right duration. There are also rapid diagnostic tests that can help diagnose a bacterial infection and avoid unnecessary antibiotic treatments (if the diagnostic tests are negative).
The proper use of antibiotics means the right indication, the right drug, the right dose, and the right duration of treatment.
Measures to Prevent Transmission
Infection prevention is the cornerstone of the fight against antibiotic resistance. Indeed, any antibiotic treatment carries a risk of bacterial resistance.
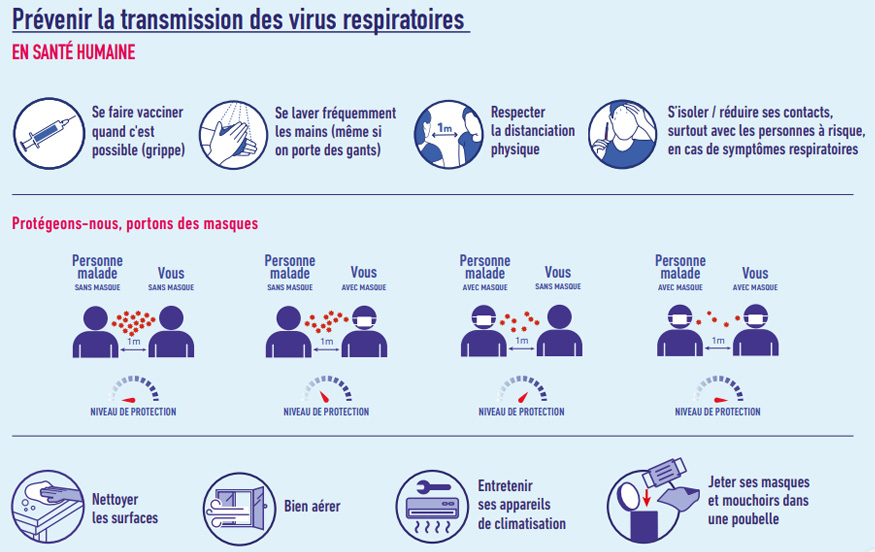
Hand hygiene and adherence to hygiene precautions are the main measures for limiting the transmission of both resistant and non-resistant bacteria.
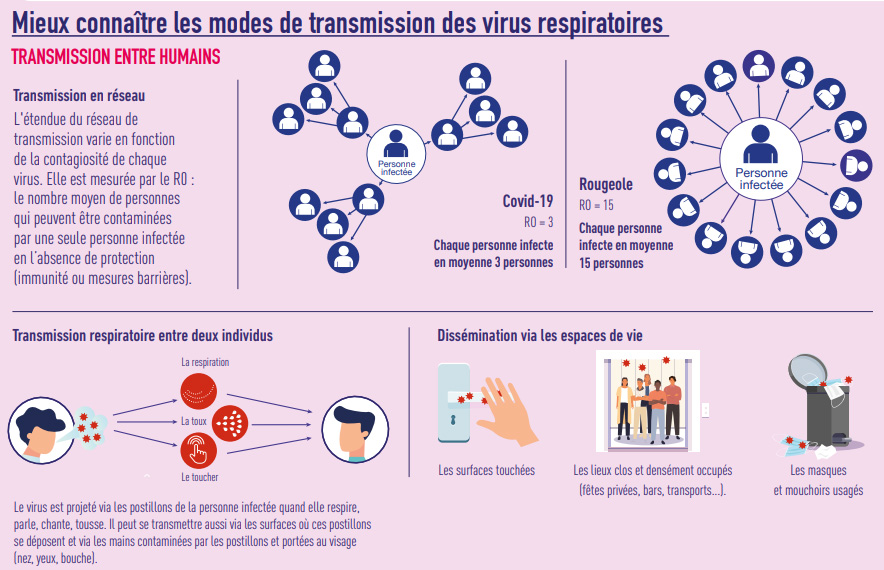
Although antibiotics are not effective against viruses, respiratory viral infections are frequently treated with antibiotics—either because of difficulties in distinguishing them from bacterial respiratory infections or due to concerns about complications from bacterial superinfections (which are, however, rare), particularly in young children and the elderly. Thus, every viral infection avoided represents one less temptation to prescribe antibiotics.
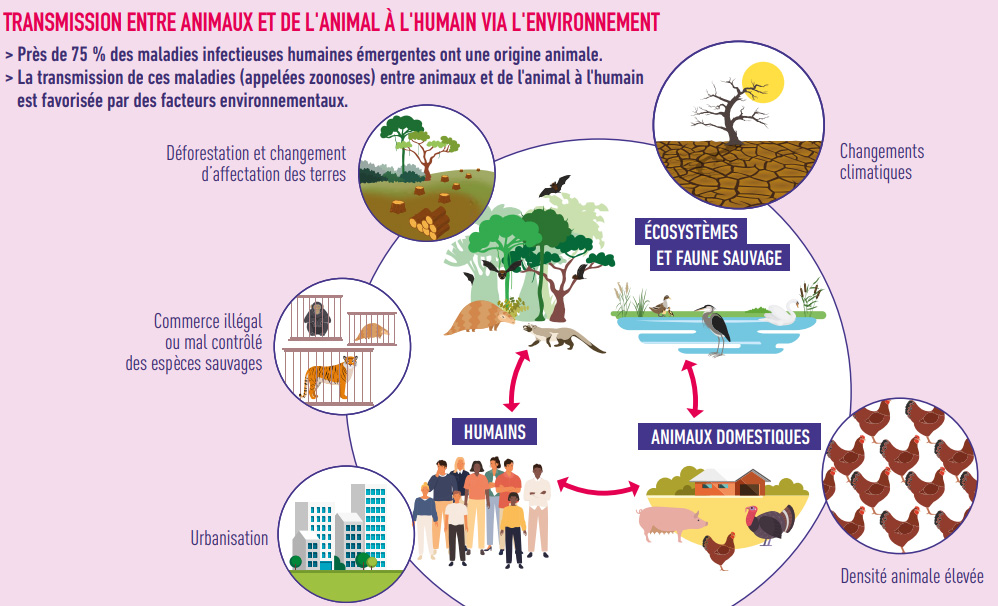
Furthermore, it is important to better understand how these infections are transmitted in order to prevent them more effectively.
Antibiotic resistance is a global threat with consequences for individuals
Antibiotic resistance affects humans, but also animals and the environment, which are interconnected and form a single whole. The fight against antibiotic resistance therefore requires a global “One Health” approach.
Antibiotic resistance threatens all life, everywhere in the world
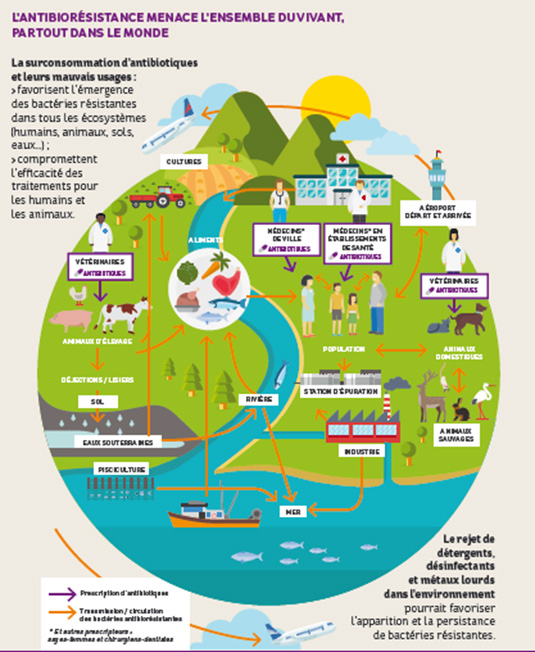
Human health, animal health, and ecosystem health are closely linked to preserving the effectiveness of antibiotics
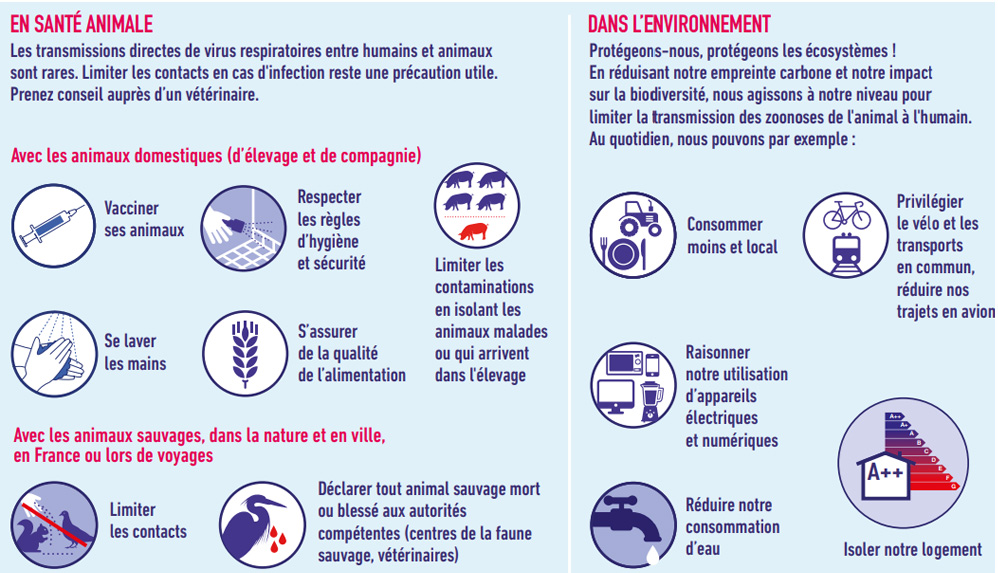
The fight against antibiotic resistance involves preventing infections through hygiene measures (with hand hygiene at the forefront), vaccination, and the prudent use of antibiotics in human and animal health.
“Individual consequences”
Today, certain bacterial resistances can complicate the treatment of an infection, even one that is initially mild. Anyone (human or animal) can, at some point, become infected with a bacterium that is multi-resistant to antibiotics, regardless of their health status and risk factors.
In this situation, the range of available treatments is limited, leading to delays in administering the most effective therapy; recovery time may be prolonged, with an increased risk of complications and mortality for the most severe infections. The antibiotics that are still effective against multidrug-resistant bacteria are second-line antibiotics, according to current guidelines, and sometimes carry a higher risk of adverse effects.
Finally, in human healthcare, these second-line antibiotics are often available only by injection, and some exclusively in hospitals, which complicates patient care, reduces patients’ quality of life, and carries risks of nosocomial infections. Fortunately, cases of infections caused by bacteria resistant to all antibiotics remain, to date, rare in France.