What are the potential consequences of carbon monoxide exposure?
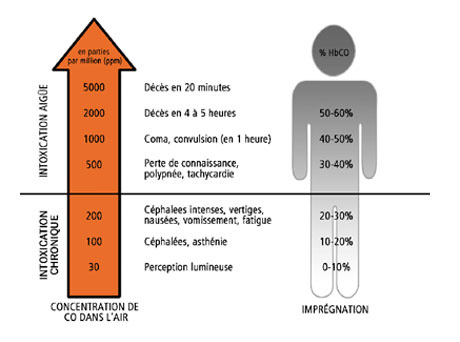
Carbon monoxide is a gas that is easily absorbed by the lungs; it then enters the bloodstream, where it competes with oxygen. In fact, carbon monoxide has an affinity for hemoglobin that is 230 times greater than that of oxygen. It therefore binds easily to hemoglobin to form a stable molecule, carboxyhemoglobin (HbCO). This phenomenon leads to a decrease in the blood’s ability to transport oxygen, greater difficulty in releasing oxygen to tissues, and asphyxia that can sometimes be fatal.
The severity of poisoning depends on the amount of carbon monoxide (CO) bound by hemoglobin. This depends on the concentration of CO in the air, the duration of exposure, and the volume of air inhaled.
Chronic CO poisoning results from repeated exposure to low concentrations over extended periods. Acute poisoning, on the other hand, follows exposure to high concentrations of carbon monoxide. It can be sudden (occurring within minutes) or develop gradually over a short period (a few hours).