Evaluation of the Organized Colorectal Cancer Screening Program for the 2018–2019 and 2020 Periods: National Indicators
National indicators
The results presented correspond to data from individuals who underwent a fecal immunochemical test (FIT) for colorectal cancer screening:
between January 1, 2018, and December 31, 2019;
between January 1, 2020, and December 31, 2020.
For the year 2020, given its exceptional nature due to the COVID-19 pandemic, a number of indicators are also presented. These are indicators related to the screening process (number of colonoscopies performed following a positive test, time to colonoscopy following a positive test, etc.). The indicators describing lesions detected during colonoscopies performed following a positive FIT test in 2020 are not presented here. This is because the reported 2020 data do not have the necessary historical perspective to yield consolidated results (see table).
A description of the indicators is available in the guide to data formats and definitions of indicators for the national program evaluation.
Quality Indicators for Screening Tests and Diagnostic Tests
National quality indicators for screening tests and diagnostic examinations are available in the table in xlsx format and pdf format for the years 2018–2019 and in the table in xlsx format and pdf format for the year 2020.
Test performance quality
2018–2019
A total of 5.2 million people underwent a screening test between January 1, 2018, and December 31, 2019, of whom 54.5% were women and 24.8% were initial screenings. Among those screened, 379,000 (7.2%) had at least one test that proved unanalyzable during the period. The proportion of people with at least one non-analysable test is higher among men (7.6%) than among women (6.9%). It is also higher for a first screening (10.9%) than for a subsequent screening (6.0%). The main reasons for non-analysable tests were: expired tube (60.7%), sample collected more than 6 days prior (17.1%), and missing collection date (14.0%). Following the non-analysable test, 279,000 people (73.5%) took another test—this time analysable—during the period. Among all screened individuals, 100,000 people (1.9%) had an unanalyzable test that was not followed by an analyzable test during the period. This rate is identical for women and men and is higher for initial screening (3.9%) than for follow-up screening (1.3%).
2020
Between January 1 and December 31, 2020, 2.9 million people underwent a screening test, of whom 54.3% were women and 21.5% were initial screenings. The proportion of women remains stable; the proportion of initial screenings continues to decline (30.9% in 2016–2017 and 24.8% in 2018–2019). Among those screened, 7.8% had at least one test that proved unanalyzable during the period, and 3.4% had an unanalyzable test that was not followed by an analyzable test performed during the period. The main reasons for non-analysable tests were: expired tube (45.9%), sample collected more than 6 days prior (36.6%), and missing collection date (11.7%).
Positive Test
2018–2019
Among the 5.1 million people with an analyzable test, 198,000 (3.8%) tested positive. This rate is higher among men (4.6%) than among women (3.1%). It is also higher for initial screenings (4.5%) than for subsequent screenings (3.6%).
2020
Among the 2.9 million people with an analyzable test, 3.6% tested positive.
Colonoscopies performed following a positive test
People who underwent a colonoscopy following a positive test
2018–2019
Among the 196,000 people eligible for a colonoscopy after a positive test, 169,000 people underwent a colonoscopy (86.4%). This colonoscopy completion rate is lower than in the 2016–2017 period (88.9%). It varies by department, with 28 departments having a rate below the acceptable European benchmark (85%) and 31 having a rate above the desirable European benchmark (90%). This percentage is higher among women (86.9%) than among men (86.0%) and for follow-up screenings (88.4%) than for initial screenings (81.5%).
National Organized Colorectal Cancer Screening Program, 2018–2019 – Percentage* of people who underwent a colonoscopy following a positive test, by department
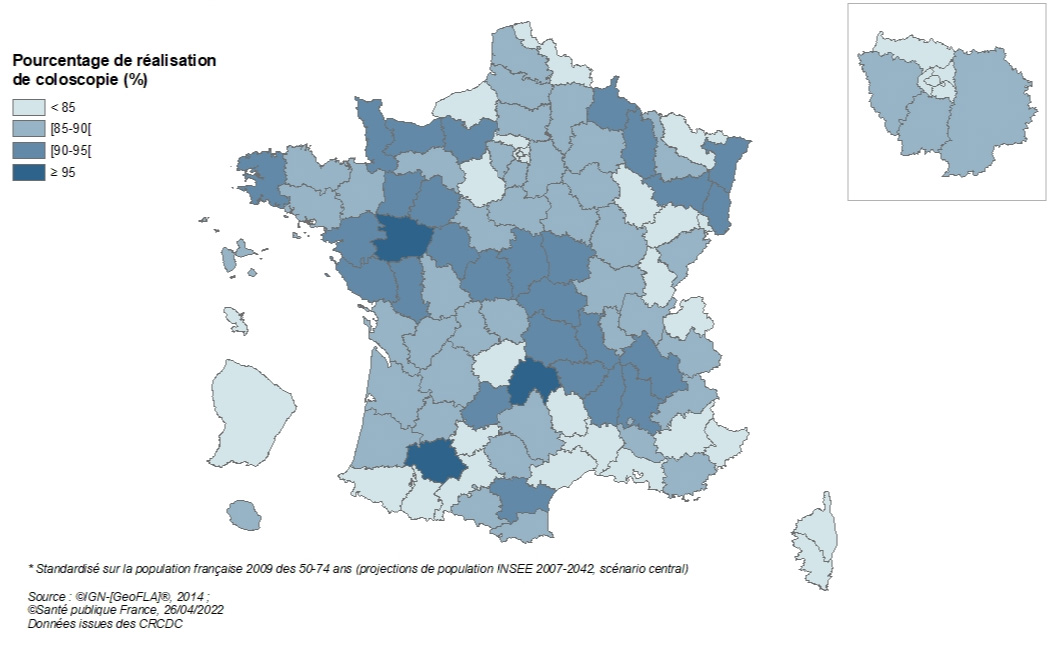
2020
Of the 103,000 people eligible for a colonoscopy following a positive test, 76.5% underwent a colonoscopy. This colonoscopy completion rate is lower than in previous periods (between 85% and 89%). While this result can be explained by the COVID-19 crisis, which led to a significant decrease in the number of colonoscopies performed in 2020, it could also be explained, at least in part, by the early collection of 2020 data (see table), which means that information regarding colonoscopies following a positive FIT test performed during the second half of the year is incomplete.
Time to colonoscopy
2018–2019
The median time between the date of the screening test result and the colonoscopy performed in the event of a positive FIT is 80 days; it is less than 3 months for 60% of individuals eligible for colonoscopy. It does not differ by sex or screening tier. It varies from 61 to 128 days depending on the department.
2020
For the 78,900 colonoscopies performed, the median time between the date of the screening test result and the colonoscopy performed following a positive FIT result is 87 days; it continues to increase compared to previous periods: 62 days in 2013–2014, 76 days in 2016–2017, and 80 days in 2018–2019. It is less than 3 months for 53% of individuals eligible for colonoscopy and ranges from 60 to 129 days for all departments except Guadeloupe, where the median wait time is 142 days.
Quality of colonoscopy preparation
2018–2019
The quality of colonoscopy preparation was rated as good for 82.7% of colonoscopies performed (82.0% among men and 83.5% among women), average for 6.6% of colonoscopies performed (7.2% among men and 6.0% among women), and insufficient for 2.0% of colonoscopies performed (2.1% among men and 1.8% among women). Information on the quality of the colonoscopy was missing for 8.7% of the exams.
A colonoscopy is considered incomplete when the entire colon and rectum are not examined during the procedure. The percentage of people who underwent a complete colonoscopy was 93.4%, similar for both men and women. It is higher for follow-up screenings (93.7%) than for initial screenings (92.6%). This figure is slightly higher than the acceptable benchmark recommended at the European level (90%) and lower than the desirable benchmark (95%). This figure has increased slightly compared to the 2016–2017 period (92.7%).
The reasons why colonoscopies were incomplete vary by sex. These include:
anatomical reasons in 32.3% of cases among men versus 38.0% among women,
obstructive lesions in 23.7% of men versus 23.2% of women,
inadequate preparation in 23.0% of men versus 20.5% of women,
an unknown reason in 21.0% of men and 18.2% of women.
2020
The quality of colonoscopy preparation was rated as good for 80.6% of colonoscopies performed, average for 6.2% of colonoscopies performed, and inadequate for 2.3% of colonoscopies performed. Information on the quality of the colonoscopy was missing for 10.8% of the exams. The percentage of people who underwent a complete colonoscopy was 91.4%.
For the following sections, only the 2018–2019 indicators are presented, as there is insufficient data to present those for 2020.
Follow-up on tests
Morbidity following colonoscopy
The completeness of data collection on colonoscopy complications, particularly deaths occurring in the days following the procedure, cannot be guaranteed at the national level. In fact, for 47% of colonoscopies, the variable recording colonoscopy incidents and accidents was not filled in, meaning it is unclear whether the information is missing or whether the colonoscopy proceeded without complications.
Nevertheless, at the national level, there were 483 cases of colonoscopy resulting in a complication (related to anesthesia, general health, or the endoscopy procedure) and 132 cases of colonoscopy resulting in a serious complication (transfusion, perforation, hospitalization lasting more than 72 hours, or death). Given the incompleteness of these data, the statistics on colonoscopy morbidity cannot be used as is.
Lost to follow-up
A person is considered lost to follow-up when the CRCDC is unable to obtain any information about that person 24 months after the positive test result. The percentage of individuals lost to follow-up is 9.6% (10.1% among men and 8.9% among women) for all departments participating in the program. This figure is higher than in the previous period (7.3% in 2016–2017).
This percentage varies by department: it is less than 3% for the departments of Yonne, Rhône, Indre, Vosges, Loire, Allier, and Finistère, and it is greater than 20% for the departments of Corsica, Paris, Hérault, Haute-Garonne, Lozère, Hautes-Pyrénées, and French Guiana. These departmental disparities reflect the heterogeneity in the quality of data collection regarding the follow-up of individuals who tested positive (for example, the reporting of information on tests performed and their results by healthcare professionals to the CRCDC is more or less systematic).
Individuals who tested positive, with whom direct or indirect contact was possible, and who were informed of the positive result but did not undergo a colonoscopy will be considered ineligible for screening after two years and following a final reminder letter.
Lesions detected
These are indicators relating to the nature and rates of lesions detected (adenomas, advanced adenomas, and cancers) through diagnostic examinations (colonoscopy or other). These indicators are listed in the table (xlsx format and pdf format).
Over the 2018–2019 period, the program detected 48,370 advanced adenomas and 11,235 colorectal cancers.
The detection rate for advanced adenomas (and colorectal cancers, respectively) is defined as the proportion of patients in whom an advanced adenoma (or cancer, respectively) is the most serious lesion detected, among those who underwent an analyzable screening test. It is expressed per 1,000 (‰) screened individuals.
The detection rate for advanced adenomas (adenomas 10 mm or larger, or with high-grade dysplasia, or a villous component) was 9.3 per 1,000 screened individuals. This is lower than the rate observed during the previous 2016–2017 period (12.3‰), which was the first period following the introduction of the immunological test. The higher sensitivity of the FIT test compared to the Hemoccult test may explain the elevated rate observed in 2016–2017 as a catch-up of lesions that had not been detected previously.
The detection rate for advanced adenomas was higher among men (12.9‰) than among women (5.9‰); it increased with age, rising among men from 9.6‰ for those aged 50–54 to 17.1‰ for those aged 70–74, and among women from 4.6‰ for the youngest to 8.1‰ for the oldest. It was also, for both men and women, higher during the first screening (11.5‰) than during subsequent screenings (8.7‰), regardless of age group (see figure below).
Figure 1 - Distribution of the detection rate of advanced adenomas (adenomas 10 mm or larger, or presenting high-grade dysplasia, or a villous component) by age, sex, and screening round, 2018–2019
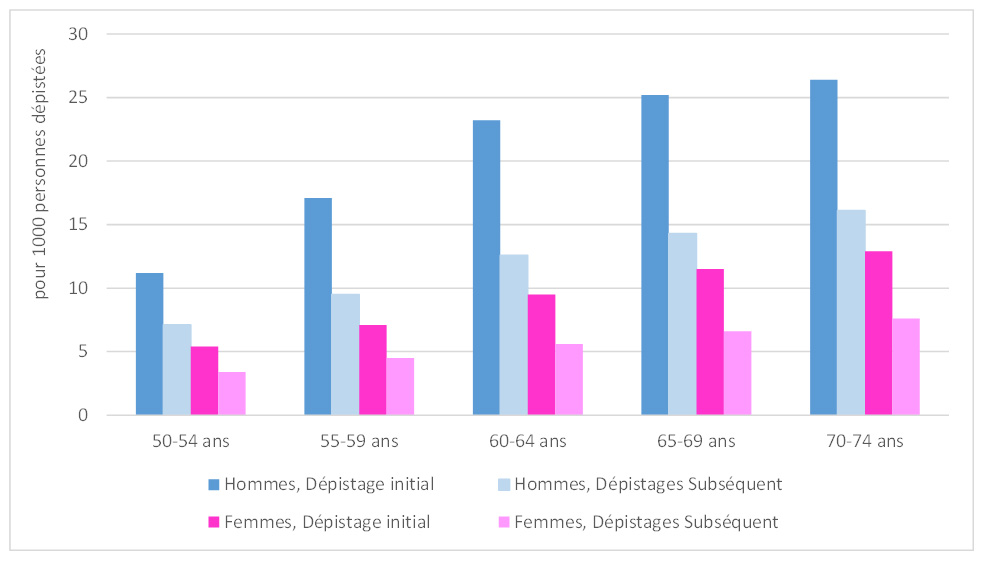
Source: Data from the CRCDC. Analysis by Santé publique France, March 2022.
The cancer detection rate was 2.1 per 1,000 people screened. This is lower than the rate observed during the previous period, 2016–2017 (3.0‰).
The cancer detection rate was higher among men (2.7‰) than among women (1.5‰); it increased with age, rising from 1.3‰ among men aged 50–54 to 4.8‰ among those aged 70–74, and from 1.0‰ among the youngest women to 2.5‰ among the oldest. It was also, for both men and women, higher during a first screening (3.0‰) than during a subsequent screening (1.9‰), regardless of age group (see figure below).
Figure 2 - Distribution of cancer detection rates by age, sex, and screening round, 2018–2019
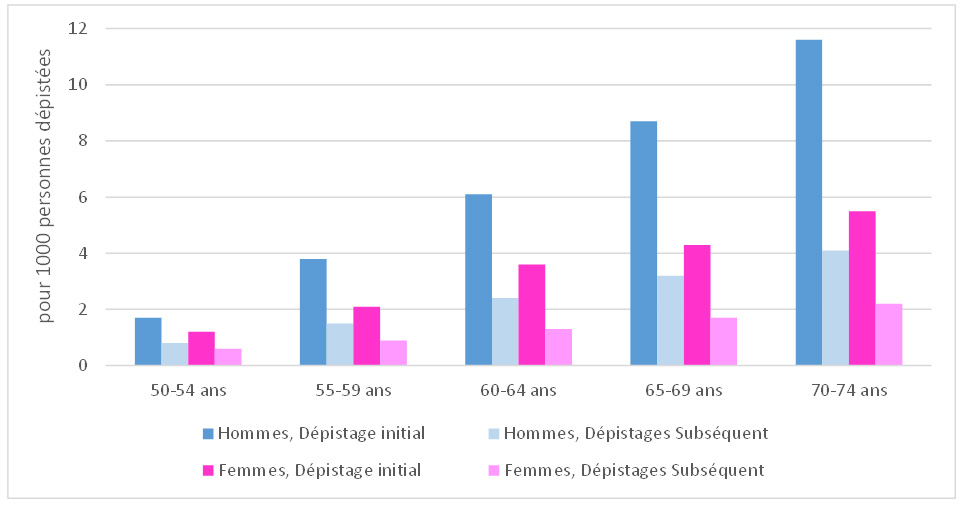
Source: Data from the CRCDC. Analysis by Santé publique France, March 2022.
Detection rates for advanced adenomas and colorectal cancers also vary by department; they depend heavily on the departmental incidence and prevalence of cancers and on the performance of departmental programs in detecting these lesions. The departmental percentages of colonoscopies performed among eligible individuals following a positive test and the percentages of individuals for whom the CRCDC was able to obtain a final diagnosis are factors influencing the recording of these lesions and may help explain departmental disparities. The definition of advanced adenomas, as defined in the “Guide to Data Format and Definitions of National Program Evaluation Indicators” and as recommended at the European level, aims to assess the proportion of lesions at high risk of malignant transformation. These are adenomas 10 mm or larger and/or presenting a villous or tubulo-villous component and/or having high-grade dysplasia. This definition differs from the definition used for clinical follow-up after polypectomy, which aims to guide colonoscopic surveillance protocols, particularly regarding the timing of recall. The lack of intra- and inter-examiner reproducibility in the diagnosis of advanced adenomas may also help explain the regional differences in observed rates of advanced adenomas.²
Screening Test Performance
The positive predictive value (PPV) of a test is the probability that a person actually has the disease if their test result is positive. It is calculated by dividing the number of lesions detected (advanced adenomas or cancers) by the number of people who tested positive and underwent a colonoscopy.
The PPV of the screening test for advanced adenomas is 28.4% and 6.6% for cancers. These PPVs are higher in men than in women (32.9% and 22.8% for advanced adenomas, and 7.1% and 6.0% for cancers, respectively) and higher for initial screenings than for subsequent screenings (31.3% and 27.3% for advanced adenomas, and 8.2% and 6.0% for cancers, respectively).
Stages of Detected Cancers
The stage of cancers detected by the organized screening program was determined according to the international TNM classification of malignant tumors.³ This classification system allows cancers to be categorized according to their degree of anatomical spread: local (or T), lymph node (or N), and metastatic (or M) involvement. For the colon and rectum, it applies only to adenocarcinomas. For colon cancers, the pTNM histopathological classification was used. For rectal cancers, which may undergo neoadjuvant treatments prior to surgery—resulting in a change in the T, N, or M stage of the detected tumor—the quality of available data and the heterogeneity of methods for documenting staging information do not allow for the determination of the pre-treatment TNM stage. Consequently, only colon cancers (including the rectosigmoid junction) and rectal cancers that did not receive neoadjuvant therapy were included.
The cancer stage is considered unknown when at least one of the T, N, or M data points is unknown (unless pT=Tis, in which case the TNM stage is in situ, or unless M=M1, in which case the TNM stage is IV). Among the detected adenocarcinomas of the rectum (without neoadjuvant treatment) and colon, this information is missing in 36.1% of patients. When the stage cannot be determined, T is missing in 17.2% of cases, N in 20.2% of cases, and M in 34.9% of cases. This percentage of adenocarcinomas with unknown stage reveals significant regional variation, as information on the metastatic status of detected cancers is difficult to access.
The description covers only the 42 departments with less than 30% missing data on cancer stages, corresponding to 5,167 detected colorectal adenocarcinomas (without neoadjuvant therapy). Among these adenocarcinomas, 11.6% are of unknown pTNM stage.
Among adenocarcinomas with a known stage, 29.5% are carcinomas in situ and 70.5% are invasive cancers (stages I to IV), of which 43.9% are stage I, 22.8% are stage II, 23.2% are stage III, and 10.1% are stage IV (see table in xlsx format and pdf format).
1See, e.g., Meyer A, Drouin J, Zureik M, Weill A, Dray-Spira R. Colonoscopy in France during the COVID-19 pandemic. Int J Colorectal Dis. 2021;36(5):1073-1075. doi:10.1007/s00384-020-03816-3.
2Denis, B., Bottlaender, J., Breysacher, G., Chiappa, P., Peter, A., Weiss, A.-M. The advanced adenoma: A topical concept but an out-of-date definition? (2011) Hepato-Gastro, 18 (3), pp. 269-276.
3Sobin M, Gospodarowicz L, Wittekind C. (eds). International Union Against Cancer. TNM Classification of Malignant Tumors, 7th Edition. Chichester: Wiley-Blackwell; 2009. 310 pages.














