Evaluation of the Colorectal Cancer Screening Program for the 2011–2012 Period: National Indicators
A description of the indicators is available in the Data Format and Indicator Definitions Guide for the National Program Evaluation
Indicators of Screening Test and Examination Quality
Reminder: The guaiac test (Hemoccult® II) was replaced by the immunoassay in April 2015.
This subsection presents screening test and examination quality indicators for the 2011–2012 period, including the rate of individuals with at least one unanalyzed test, the rate of individuals with an unanalyzed test that was not retested, the rate of positive tests, the percentage of colonoscopies performed…
The national quality indicators for screening tests and examinations are shown in the table opposite: xls format and pdf format
Additional information:
According to European recommendations1
the percentage of colonoscopies performed following a positive test
the acceptable level is 85%
the desirable level is 90%
the percentage of complete colonoscopies (entire colon and rectum examined):
the acceptable level is 90%
the target rate is 95%
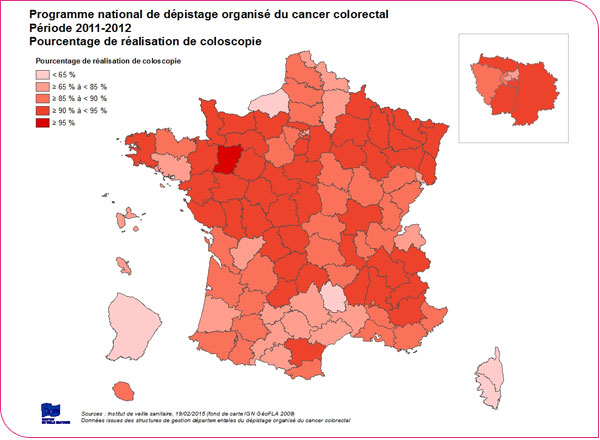
Over the 2011–2012 period, the percentage of colonoscopies performed following a positive test result was 86.6%, fairly close to the recommended European level, with significant regional disparities. It is 90% or higher in 46 departments, but remains low (between 25% and 65%) in 4 departments (Corsica, Seine-Maritime, French Guiana, Lozère). The low rate of colonoscopy completion observed in certain departments is likely linked to patients’ acceptance of the test, under-resourcing of medical services in some areas—resulting in longer wait times—and the incomplete reporting of this data to administrative bodies, likely due to inconsistent engagement by healthcare professionals.
Percentage of people who underwent a colonoscopy following a positive test
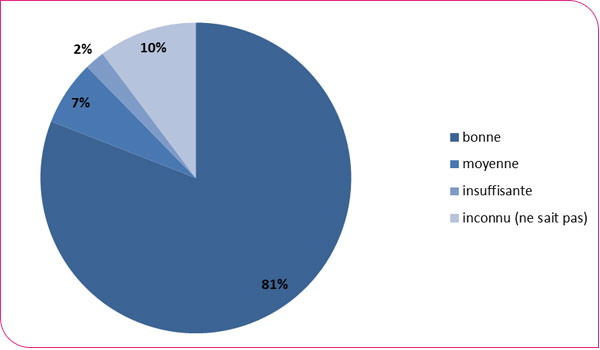
Distribution of colonoscopies performed by quality of preparation
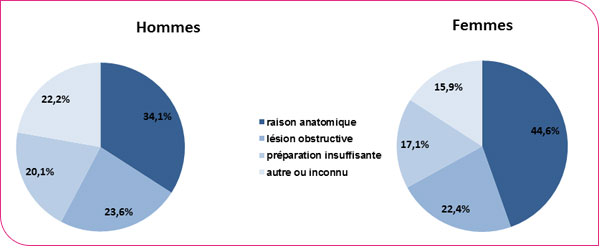
Distribution of incomplete colonoscopies by reason
Colonoscopy morbidity
The completeness of data collection on colonoscopy complications, particularly deaths occurring in the days following the procedure, is not ensured at the national level. The variable tracking colonoscopy incidents and accidents contains 54.2% of unknown data, making it impossible to determine whether the information is missing or if the colonoscopy proceeded without complications. These statistics therefore cannot be presented. More detailed analyses need to be conducted on this topic.
1 Segnan N, Patnick J, von Karsa L (eds). European guidelines for quality assurance in colorectal cancer screening and diagnosis. First Edition. Luxembourg: European Commission, 2010.
Program monitoring indicators
This subsection presents program monitoring indicators: the time taken to perform a colonoscopy and the percentage of lost-to-follow-up cases for the years 2011–2012.
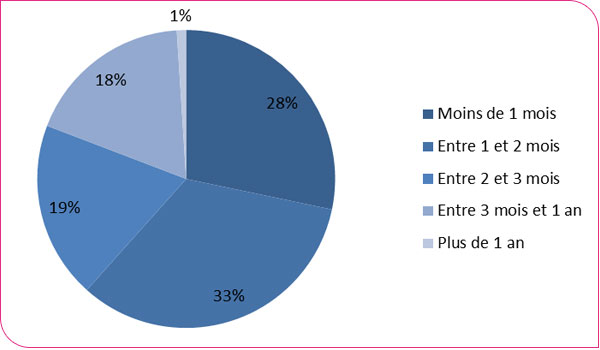
Distribution of time to colonoscopy
The distribution of wait times for colonoscopies following a positive test shows that only 28% of colonoscopies are performed within 31 days, in accordance with European recommendations (Figure 3). The median turnaround time is 57 days [interquartile range (IQR): 38 to 87], and varies considerably by department, ranging from 37 days (IQR: 23–65) in the Alpes-Maritimes to 87 days (IQR: 58 to 129) in the Vienne department (xls format and pdf format). While these delays may reflect the degree of motivation among those who tested positive to continue the diagnostic process, they also reflect the waiting times for obtaining an appointment. This significant delay can be a source of additional anxiety for beneficiaries
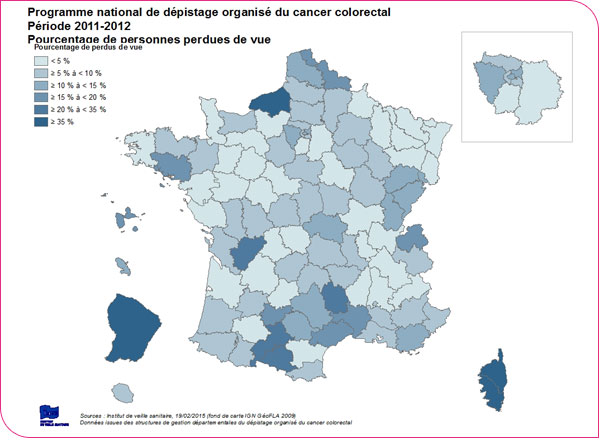
Geographic distribution of the percentages of patients lost to follow-up
A person is considered a "lost to follow-up" case when the managing organization is unable to obtain any information about that patient 24 months after the test was performed. As of April 30, 2014, the percentage of "lost to follow-up" cases was 8.6% (8.9% among men and 8.2% among women) for all 99 departments participating in the program, and varies by department (from 0% in the Ardennes and Meuse departments to 50.7% and 53.7%, respectively, in the Corsica and Seine-Maritime departments). It is also high for the departments of Charente (26.6%), Haute-Garonne (26.7%), and French Guiana (36.3%). These departmental disparities reflect the heterogeneity of documentation within management structures regarding test follow-up (for example, the transmission of information by healthcare professionals to the management structure is more or less systematic).
Indicators of detected lesions
This subsection presents indicators of detected lesions. These include indicators regarding the nature of lesions detected by diagnostic examinations (colonoscopy or other) and lesion detection rates (adenomas, advanced adenomas, and cancers).
The detection rate for advanced adenomas (and colorectal cancers, respectively) is defined as the proportion of patients in whom an advanced adenoma (or cancer, respectively) is the most serious lesion detected, among those who underwent an analyzable screening test. It is expressed per 1,000 people screened.
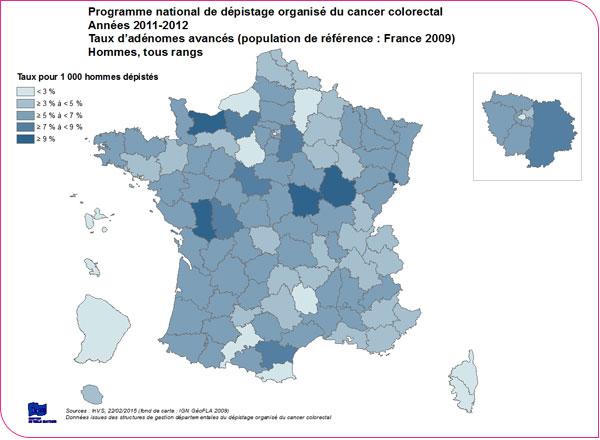
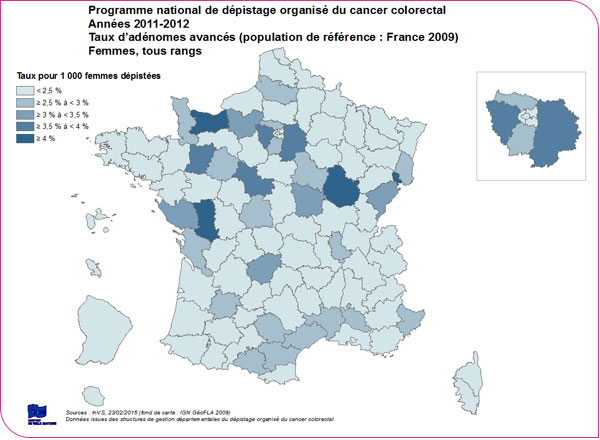
An advanced adenoma (defined as an adenoma 10 mm or larger, or one with high-grade dysplasia, or a villous component) is detected in 3.8 people per 1,000 screened (xls format and pdf format). This rate is higher among men than among women (5.3‰ versus 2.3‰) and increases with patient age: for patients under 55 years of age, it is 3.7‰ for men and 1.6‰ for women, whereas for patients aged 70 to 74, it is 7.3‰ for men and 3.4‰ for women.
There is significant regional variation, with detection rates for advanced adenomas ranging from 0.3‰ in Corsica to 11.4‰ in Côte-d'Or among men, and from 0.3‰ to 5.3‰ among women, respectively (XLS format and PDF format).
The cancer detection rate is 1.4 per 1,000 people screened, with variations by age, sex, and screening round. Among women, it is 1.1‰ during the first screening and 0.9‰ during subsequent screenings, whereas among men, it is 2.2‰ and 1.9‰ respectively—twice as high (xls format and pdf format). It also varies by department (xls format and pdf format).
The detection rates for advanced adenomas and colorectal cancers depend heavily on the departmental incidence of cancer and the performance of departmental programs in detecting these lesions. Indeed, the percentage of eligible individuals who undergo a colonoscopy following a positive test result and the percentage of individuals for whom the managing facility lacks a final diagnosis (lost to follow-up or final diagnoses unknown) are factors that influence the recording of these lesions and may help explain departmental disparities. Furthermore, as defined in the "Guide to Data Format and Definitions of National Program Evaluation Indicators," advanced adenomas are adenomas 10 mm or larger in size, with villous or tubulo-villous components, or with high-grade dysplasia. The lack of intra- and inter-examiner reproducibility in the diagnosis of advanced adenomas helps explain the departmental differences in observed rates of advanced adenomas.


Indicators of Detected Cancers
The stage of cancers detected by the organized screening program was determined according to the TNM classification system for malignant tumors1. Since this classification applies only to carcinomas of the colon and rectum, only cancers histologically classified as “adenocarcinoma” are described in this section. For colon cancers, the pTNM histopathological classification was used. For rectal cancers, which may undergo neoadjuvant treatments prior to surgery—resulting in a change in the T or N stage of the detected tumor—the quality of the available data and the heterogeneity of the methods used to document staging information do not allow for the determination of the pre-treatment TNM stage. Consequently, only colon cancers (including the rectosigmoid junction) and rectal cancers that did not receive neoadjuvant therapy were included (N = 6,381).
The cancer stage is considered unknown when at least one of the T, N, or M values is unknown (unless pT=Tis, in which case the TNM stage is in situ, or unless M=M1, in which case the TNM stage is IV). Of the 6,381 rectal (without neoadjuvant therapy) and colon adenocarcinomas detected, this information is missing for 24.9% of patients. This national percentage reflects significant regional variation (Percentage of unknown stage: xls format and pdf format), as information on the metastatic status of detected cancers is difficult to obtain.
The description covers only the 58 departments with no more than 30% of missing data on cancer stages, corresponding to 4,370 detected adenocarcinomas of the colon and rectum (without neoadjuvant therapy). Among these adenocarcinomas, 9.2% are of unknown pTNM stage.
Among adenocarcinomas with known stages, 26% are carcinomas in situ and 74% are invasive cancers (stages I to IV), of which 37.6% are stage I, 27.3% are stage II, 24.0% are stage III, and 11.1% are stage IV (xls format and pdf format).














