The disease
An arbovirus with an evolving and unpredictable epidemiology
West Nile Virus is an arbovirus of the genus Flavivirus (family Flaviviridae). It was first isolated in 1937 in the West Nile District of Uganda, hence its name. It has historically been responsible for epidemics in Africa, the Middle East, Western Asia, and Europe. It first appeared in North America in 1999 and spread throughout the entire region within a few years.
In mainland France, it is regularly detected along the Mediterranean coast.
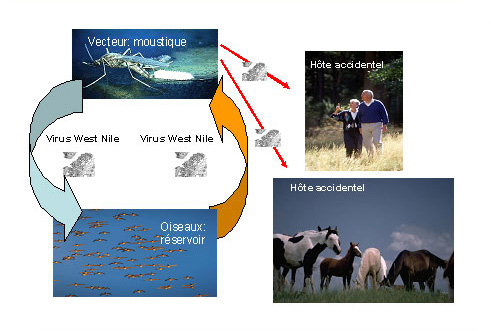
It is a bird virus that can infect humans and horses, which are “accidental hosts.” There are several lineages of this virus.
The epidemiology of the West Nile virus, influenced by numerous factors—particularly environmental ones—is dynamic and difficult to predict.
In Europe, the virus was initially introduced by migratory birds from Africa. It is now established and endemic in several areas of southern and central Europe. The introduction by migratory birds is combined with gradual endemicization. Since 2010, increased circulation has been observed in Europe, Russia, and the Mediterranean region. The year 2018 was marked by the largest epidemic on record; the number of cases reported in Europe exceeded the total from the previous seven years combined. In 2019 and 2020, human cases were reported for the first time in Germany (2019 and 2020) and the Netherlands (2020).
Virus lineage 2, first detected in a bird in Hungary in 2004, is gradually spreading across Europe and now coexists with lineage 1, which was historically the only one present in Europe.
In this context, Santé publique France’s priorities are:
To detect human cases in France as early as possible
To contribute to the multidisciplinary surveillance of this virus in collaboration with veterinary health stakeholders and entomologists
To adapt the surveillance strategy to epidemiological developments
To contribute to the implementation of preventive measures, particularly regarding the safety of products of human origin (labile blood products, tissues, cells, and organs)
To help inform healthcare professionals, local authorities, and the general public about protective measures against mosquitoes.
A zoonosis transmitted primarily by mosquitoes
Birds are the primary reservoir of the West Nile virus, but humans, horses, and other vertebrate species are also susceptible to infection. Humans and horses are accidental hosts because the transmission cycle occurs between birds and bird-biting mosquitoes.
Transmission of the West Nile virus to humans occurs primarily through a mosquito bite. This is mainly from mosquitoes of the Culex genus. The mosquito becomes infected by feeding on infected birds. A mosquito infected in this way can, upon biting another host, transmit the virus to another bird or to an accidental host such as a human or a horse.
Humans and horses represent "epidemiological dead ends" for the West Nile virus because the amount of virus in the blood (viremia) is insufficient to infect the mosquito during a bite and thus allow transmission of the disease.
In temperate zones, WNV transmission is seasonal, occurring during the mosquito activity period (from June to late November in mainland France).
The virus can also be transmitted, more rarely, through human-derived products: blood transfusions and organ, tissue, or cell transplants. Cases of mother-to-child transmission during pregnancy, childbirth, and breastfeeding have also been reported.
A disease that is usually asymptomatic but can be serious
While 80% of infected individuals remain asymptomatic or have only mild symptoms, the West Nile virus can cause neurological complications (meningitis, encephalitis, and meningoencephalitis), which account for its severity. These neuroinvasive forms are more common in older adults and can lead to long-term complications and even be fatal in humans.
In 20% of infected individuals, a flu-like syndrome develops (sudden onset of fever, headache, joint and muscle pain) that may be accompanied by a rash.
Severe forms of the disease occur in fewer than 1 in 100 infected individuals, primarily adults and particularly the elderly. These are neuroinvasive forms manifesting as meningitis, meningoencephalitis, flaccid paralysis, or Guillain-Barré syndrome.
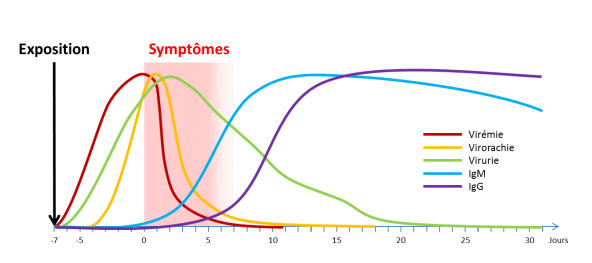
The incubation period typically lasts 2 to 6 days but can range from 2 to 14 days.
The diagnosis can be confirmed by laboratory tests performed on blood and/or cerebrospinal fluid (CSF) obtained via lumbar puncture:
Serology (detection of anti-West Nile virus antibodies). IgM antibodies are typically detected 3 to 8 days after the onset of clinical symptoms and generally persist for 1 to 3 months. IgG antibodies appear 2 to 3 weeks after the onset of infection and persist for years. Two samples taken 2 to 3 weeks apart are required; a significant increase in antibody titers between these samples will confirm that the infection is recent.
Cross-reactions are observed with other flaviviruses (e.g., dengue or Zika viruses). These cross-reactions require confirmation of any positive result via a neutralization test.
Genomic amplification by RT-PCR. The viral genome is detectable in plasma 2 to 18 days after infection, or up to 5 days after the first symptoms appear.
Viral isolation by viral culture. This technique is labor-intensive and requires a Biosafety Level 3 laboratory. It is not a routine test.
Symptomatic treatment
There is no specific antiviral treatment for West Nile virus. Management therefore focuses on treating symptoms as they arise.
Severe cases are treated in a hospital setting, in the intensive care unit if necessary.
When human cases are detected, it is necessary to remind people living near the transmission site(s) of the measures to protect against mosquitoes.
Mosquito prevention measures
There is currently no commercially available vaccine for humans, nor is there a preventive treatment against West Nile virus infection. For horses, an equine vaccine is available.
Prevention of West Nile virus infection relies on mosquito protection measures, including both:
Individual: protective clothing, repellents, mosquito coils, electric diffusers, mosquito nets
And community-wide: control of larval breeding sites and, depending on the entomological situation, possible measures against adult mosquitoes.
Specific measures are in place to ensure the safety of blood transfusions and organ and tissue donations. These measures are based on screening donors or excluding high-risk donors. These measures are implemented in accordance with the recommendations issued by the High Council for Public Health (HCSP) and its permanent working group on the Safety of Human Body Components and Products (SECPROH).
Download