Salmonellosis Outbreak Caused by Salmonella enterica Serotype Agona Among Infants in France – Update as of January 12, 2018
Between August and December 2017, several infants were identified by the National Reference Center for Salmonella as having salmonellosis caused by Salmonella serotype Agona. The parents of these infants reported that their children had consumed the following brands of infant formula: Pepti Junior by Picot, Picot SL, Picot Anti-Colic, Picot Rice, and Milumel Bio 1 (palm oil-free). Santé publique France is issuing an update on the ongoing investigations and reiterating the hygiene guidelines to follow when preparing infant formula. To date, the CNR has not identified any cases of Salmonella Agona infection in infants whose symptoms began after December 2, 2017 (the date of the initial product recalls and withdrawal measures).

Update on the Situation
In late November 2017, the National Reference Center (CNR) for Salmonella (Institut Pasteur, Paris) identified an unusual number of young children with salmonellosis caused by Salmonella serotype Agona. The identification of 8 cases in 8 days triggered an alert, and epidemiological investigations began on December 1 among the parents of the affected infants.
The epidemiological investigation is being conducted by Santé publique France in collaboration with the NRC, the Directorate General for Competition, Consumer Affairs, and Fraud Control (DGCCRF), and the Directorate General for Health (DGS). To date, the investigation has identified 37 infants (16 boys and 21 girls, median age 4 months (min: 2.5 weeks, max: 9 months)) with Salmonella Agona serotype salmonellosis, which occurred between mid-August and December 2, 2017.
All of these strains belong to the same epidemic clone. One of the cases was identified retrospectively by the CNR and developed symptoms in late April 2017 (Figure 1). These 37 infants reside in 10 different regions (10 in Auvergne-Rhône-Alpes, 6 in Île-de-France, 4 in Bourgogne-Franche-Comté, 3 in Pays de la Loire, 3 in Occitanie, 3 in Nouvelle-Aquitaine, 2 in Provence-Alpes-Côte d’Azur, 3 in Centre-Val-de-Loire, 2 in Hauts-de-France, 1 in Normandy) (Figure 2).
Figure 1 - Distribution of S. Agona (epidemic clone) infection cases by week of diagnosis at the CNR, France, April–December 2017 (n=37)
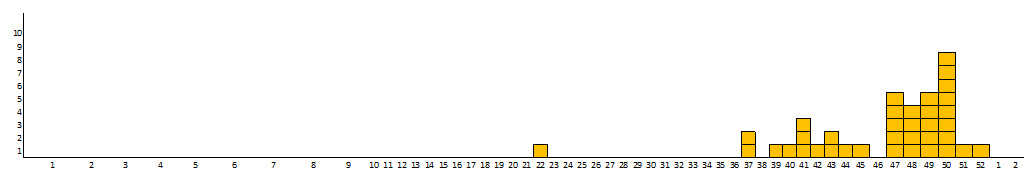
To date, the families of 36 infants have been interviewed. Among them, 18 were hospitalized due to salmonellosis; all have been discharged from the hospital. During telephone interviews, parents reported that their babies were doing well.
Of these 36 infants, 35 consumed, within 3 days prior to the onset of their symptoms, the following brands of infant formula: Pepti Junior by Picot (26 infants), Milumel Bio 1 palm oil-free (5 infants), Picot Rice (2 infants), Picot SL (1 infant), and Picot Anti-Colic (1 infant); these 5 formulas are manufactured at the same production site. One mother reported exclusively breastfeeding her infant.
Figure 2 - Geographic distribution of the 37 infants with Salmonella Agona serotype (epidemic clone) salmonellosis, France, April–December 2017
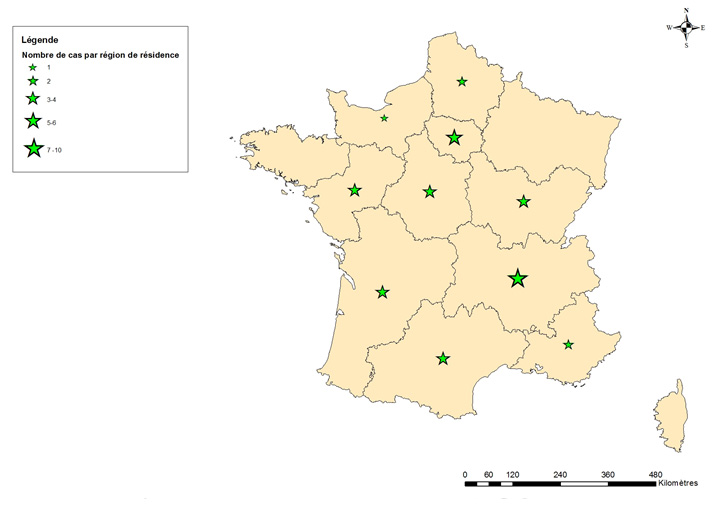
The results of the investigation strongly suggest that Picot’s Pepti Junior, Picot SL, Picot Rice, Picot Anti-Colic, and Milumel Bio 1 palm oil-free formulas, all produced at the same site, are the source of these Salmonella Agona infections.
The cases identified during this investigation involve infants who experienced diarrhea following consumption of these milk formulas, who underwent medical consultation and stool culture (stool analysis), and from whom laboratories isolated a strain of Salmonella that was submitted to the CNR. It is possible that some cases were not identified. Nevertheless, these unreported cases are generally less severe, and their number is likely limited due to more systematic medical care for infants and for severe infections.
Microbiological investigations
Based on genomic analysis of the strains, the CNR identified an epidemic clone among the S. Agona strains received in 2017.
Given the international distribution of these milk products, the CNR has published the sequence of the epidemic strain on EBI-ENA under the number ERR2219379. The national reference centers and focal points in the affected countries have been informed so that they can compare their S. Agona strain sequences with that of the French outbreak.
The epidemic strain has specific biochemical characteristics recognizable by laboratory biologists: it produces neither H2S nor gas on Kligler-Hajna medium, which is unusual for non-typhoid Salmonella.
Furthermore, this strain does not exhibit any particular antibiotic resistance.
Management Measures
Several consecutive measures involving the withdrawal and recall of a wide range of milk products manufactured by the same company have been taken since December 2, 2017. The list of these products is available on the DGCCRF website.
The DGS set up a toll-free hotline from December 10 to 24, 2017. This hotline was designed to answer parents’ questions and provide appropriate health recommendations.
The manufacturer has also made a toll-free number available to parents—0800 120 120 (open from 9:00 a.m. to 8:00 p.m.). This hotline is designed to answer parents’ questions about the recalled products.
International Investigations
As early as December 6, 2017, Santé publique France and the CNR alerted their counterparts at national public health agencies and institutes, as well as microbiologists at the European Union’s national reference laboratories, via the Epidemic Intelligence System (EPIS) of the European Centre for Disease Prevention and Control (ECDC). A report on the French epidemiological and microbiological investigations was published in the journal Eurosurveillance on January 11, 2018.
The World Health Organization (WHO) also transmitted the alert via its Infosan network to the national focal points of all potentially affected countries.
Information on the products to be withdrawn from sale was disseminated internationally by the DGCCRF via the European Rapid Alert System for Food and Feed (RASFF) and the WHO’s Infosan network.
To date, two cases of Salmonella Agona infection in infants who consumed milk from the list of implicated products have been identified outside France: one child in Spain (infected by a strain belonging to the epidemic clone) and one child in Greece (confirmation of the strain’s affiliation with the epidemic clone is pending).
The ECDC and the European Food Safety Authority (EFSA), in close collaboration with French health authorities and Santé publique France, are currently conducting a rapid risk assessment of this event. The report of this assessment will be published shortly on the ECDC website (scheduled for January 17, 2018).
Advice for Parents
Parents who are using a can of infant formula affected by this recall must switch to a different formula immediately. The French Society of Pediatrics (SFP) has issued recommendations for possible substitutes, which are also published on the website of the Ministry of Solidarity and Health. These recommendations have also been shared with general practitioners, pediatricians, midwives, and pharmacists.
Santé publique France reminds the public of the hygiene guidelines to follow when preparing baby bottles:
hands must be washed thoroughly with soap and water before handling the formula;
bottles should not be prepared in advance;
bottles must be cleaned immediately after use.
Reminders about the disease
Salmonella infections generally occur within 3 days of ingestion and cause symptoms of gastroenteritis, including vomiting, diarrhea (which may be bloody), and fever in most cases. If these symptoms appear in an infant, families should consult a doctor immediately.
Salmonella Agona is one of approximately 2,000 Salmonella serotypes that are pathogenic to humans. From 2012 to 2016, the National Reference Center for Salmonella identified approximately 65 strains of Salmonella Agona per year across all age groups. Like most other Salmonella serotypes, Salmonella Agona is found in France in various animal reservoirs, including poultry, cattle, pigs, and animal feed.
Several outbreaks of Salmonella Agona salmonellosis have been reported in the past, notably an outbreak attributed to the consumption of powdered milk by infants in France in 2005, an outbreak among infants in Germany attributed to the consumption of fennel- and anise-based herbal teas³, and outbreaks attributed to the consumption of peanut snacks⁴ or oat-based cereals⁵.
References1Jourdan-da Silva Nathalie et al. Ongoing nationwide outbreak of Salmonella Agona associated with internationally distributed infant milk products, France, December 2017. Euro Surveill. 2018;23(2):pii=17-00852. 2Brouard C, Espie E, Weill FX, Brisabois A, Kerouanton A, Michard J, Hulaud D, Forgues AM, Vaillant V, de Valk H. Salmonellosis outbreak caused by Salmonella enterica serotype Agona linked to the consumption of infant formula, France, January–May 2005. Bull Epidemiol Hebd 2006;(33):248–50. 3Koch J, Schrauder A, Werber D, Alpers K, Rabsch W, Prager R, Broll S, Frank C, Roggentin P, Tschäpe H, Ammon A, and Stark K. A nationwide outbreak of Salmonella Agona in infants due to aniseed in herbal tea, Germany, October 2002–July 2003. 5th World Congress, Foodborne Infections and Intoxications, June 7–11, 2004, Berlin, Germany.4Killalea D, Ward LR, Roberts D, et al. International epidemiological and microbiological study of an outbreak of Salmonella Agona infection from a ready-to-eat savory snack — I: England and Wales and the United States. BMJ 1996;311-13.5From the Centers for Disease Control and Prevention. Multistate outbreak of Salmonella serotype Agona infections linked to toasted oats cereal—United States, April–May 1998. JAMA. 1998 Aug 5;280(5):411.














