Cases of Salmonella Poona in Infants: Update as of February 18, 2019
On January 23, 2019, January 24, 2019, and February 8, 2019, Santé publique France issued alerts regarding cases of salmonellosis caused by the Poona serotype in infants.
To date, the National Reference Center (NRC) for Salmonella at the Pasteur Institute has identified 12 cases of Salmonella Poona (S. Poona) infection in infants, whose strains belong to the same genomic cluster (genetically related strains). In addition, the CNR has identified 14 other infants who have had Salmonella Poona (S. Poona) infection. The Salmonella strains from these 14 infants are currently being analyzed to determine whether they belong to the same genomic cluster.
Santé publique France is investigating these cases of salmonellosis by interviewing the parents of the sick infants about symptoms and food consumption prior to the onset of symptoms.
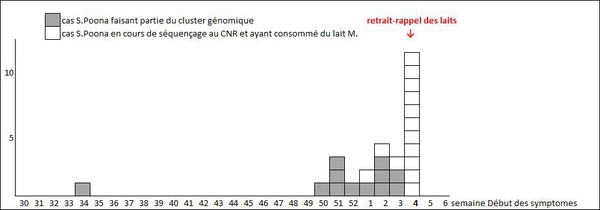
These 26 infants—18 boys and 8 girls—residing in 10 different regions in mainland France, aged 2 months to 2 years at the time of symptom onset, became ill between late August 2018 and January 27, 2019 (Figure 1). All presented with diarrhea, including 13 with blood in their stools, and 25 had a fever. Twelve infants were hospitalized for salmonellosis and have since been discharged. All children are doing better or have recovered.
Figure 1: Distribution of the 26 cases of Salmonella Poona infection, 2018–2019, France
Investigations with the parents of these 26 infants revealed that, in the days leading up to the onset of symptoms, they had consumed Modilac brand infant formula produced at the same factory in Spain: Modilac Expert Rice Stage 1, Modilac Rice Stage 2, and Modilac Rice Stage 2 Anti-Regurgitation. The 26 infants did not share any other food or drink in common. The bottles were prepared using bottled water from seven different brands.
The results of the investigation strongly suggest that Modilac Expert Rice Stage 1, Modilac Rice Stage 2, and Modilac Rice Stage 2 Anti-Regurgitation are the source of these Salmonella Poona infections.
Consequently, on January 24, 2019, Sodilac, the company marketing these products, in coordination with the Directorate General for Competition, Consumer Affairs, and Fraud Control (DGCCRF) and the Directorate General for Health (DGS), proceeded to withdraw and recall its entire line of rice protein-based infant nutrition products and, more broadly, all products manufactured at the affected Spanish production site. Sodilac has also set up a toll-free number, 0800 800 970, to answer parents’ questions.
The affected products, distributed to consumers exclusively through pharmacies and several charitable organizations, or sold on online retail sites, are being recalled regardless of batch number or manufacturing date.
On January 25, 2019, as a precautionary measure, Lactalis initiated a recall of the Picot AR product, the only product manufactured at the same Spanish facility as Modilac formulas.
Parents who still have cans from these batches should not use them, whether they are new or already opened. They should contact their pediatrician or general practitioner to receive specific advice on a replacement milk, particularly for babies allergic to milk proteins. In the meantime, they can visit a pharmacy where a substitute milk will be offered to them.
The French Society of Pediatrics has issued recommendations for possible substitutes, which are available online on the website of the Ministry of Solidarity and Health. These recommendations have also been shared with general practitioners, pediatricians, midwives, pharmacists, healthcare facilities, maternal and child health centers, daycare centers, and relevant associations.
Santé publique France takes this opportunity to remind the public of the hygiene guidelines to follow when preparing baby bottles:
hands must be washed thoroughly with soap and water before handling the bottle,
bottles should not be prepared in advance,
bottles must be cleaned immediately after use.
Salmonella infections occur within 7 days (most often within 3 days) of ingestion and cause symptoms of gastroenteritis, including vomiting, diarrhea (sometimes bloody), and fever in most cases. If these symptoms appear in an infant, families should consult a doctor.
Salmonella Poona is one of approximately 2,000 Salmonella serotypes that are pathogenic to humans. From 2016 to 2018, the National Reference Center for Salmonella identified approximately 50 strains of Salmonella Poona per year. Like most other Salmonella serotypes, Salmonella Poona is found in France in various animal reservoirs, including poultry, dairy products, and animal feed. This serotype is also found in reptiles.
Several outbreaks of Salmonella Poona-related salmonellosis have been reported in the past, notably an outbreak attributed to the consumption of powdered milk by infants in Spain in 2010–2011 (1). The Spanish factory manufacturing this powdered milk was the same as the one involved in the current outbreak in France. The S. Poona strains identified in these two outbreaks are genetically linked.
Other outbreaks of S. Poona salmonellosis have occurred in the United States: in 2015 associated with the consumption of cucumbers (2) and contact with small turtles (3), and in 2000–2002 associated with the consumption of melons (4).
References
1: Community-Wide Outbreak of Salmonella Poona Gastroenteritis in 2010–2011: http://revista.isciii.es/index.php/bes/article/view/339/3672: Multistate Outbreak of Salmonella Poona Infections Linked to Imported Cucumbers: https://www.cdc.gov/salmonella/poona-09-15/3: Four Multistate Outbreaks of Human Salmonella Infections Linked to Small Turtles: https://www.cdc.gov/salmonella/small-turtles-10-15/index.html4: Multistate Outbreaks of Salmonella Serotype Poona Infections Associated with Eating Cantaloupe from Mexico --- United States and Canada, 2000–2002: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5146a2.htm